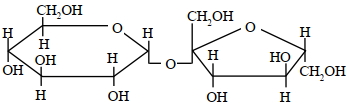
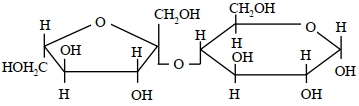
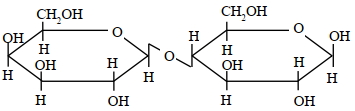
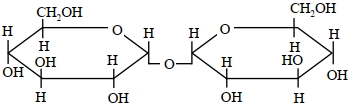
A disaccharide $X$ cannot be oxidised by bromine water. The acid hydrolysis of $X$ leads to a laevorotatory solution. The disaccharide $X$ is
IIT 2023, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich of the following gives a positive test with ninhydrin?
- 2View SolutionFor osazone formation, the effective structural unit necessary is
- 3View SolutionMaltose contains how many oxygen atoms
- 4$D\, (+)$ glucose reacts with hydroxylamine and yields an oxime. The structure of the oxime would beView Solution
- 5View SolutionWhich of the following statement is not correct?
- 6View SolutionGlucose and fructose are ......................... of each other
- 7View SolutionIdentify correct statement for sucrose
- 8Which of the following is correct structure of $\alpha-$ anomer of maltose ?View Solution
- 9View SolutionAt iso-electric point
- 10View SolutionWhich of the following is a non-reducing sugar?