Download our appand get started for free
Similar Questions
- 1View SolutionProtein can be most easily removed from
- 2Glucose when heated with $C{H_3}OH$ in presence of dry $HCl$ gas gives $\alpha $ and $\beta - {\rm{methyl}}$ glucosides because it containsView Solution
- 3The Fischer presentation of $D$-glucose is given below. The correct structure(s) of $\beta$-$L$-glucopyranose is (are)View Solution

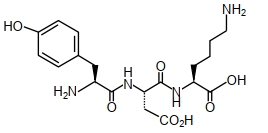
- 4The structure of a peptide is given belowView Solution
If the absolute values of the net charge of the peptide at $pH =2, pH =6$, and $pH =11$ are $\left| z _1\right|,\left| z _2\right|$ and $\left|z_3\right|$, respectively, then what is $\left|z_1\right|+\left|z_2\right|+\left|z_3\right|$ ?
- 5$D-$ Glucose and $L-$ Glucose areView Solution
- 6The compound $I$ and $II$ are related asView Solution
$\mathop {\begin{array}{*{20}{c}}
{\begin{array}{*{20}{c}}
{COOH\,\,} \\
{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,} \\
{H - C - OH\,\,\,\,\,\,\,\,\,\,\,} \\
{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}
\end{array}} \\
{Br - C - H\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,} \\
{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,} \\
{\,\,\,COOC{H_3}}
\end{array}}\limits_{(I)} $ $\mathop {\begin{array}{*{20}{c}}
{\begin{array}{*{20}{c}}
{\,\,\,\,\,\,\,\,COOC{H_3}\,\,} \\
{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,} \\
{H - C - Br\,\,\,\,\,\,\,\,\,\,\,} \\
{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}
\end{array}} \\
{HO - C - H\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,} \\
{\,\,|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,} \\
{\,\,\,\,\,\,\,COOH\,\,\,\,\,\,\,\,}
\end{array}}\limits_{(II)} $ - 7View SolutionWhich of the following statements is correct ?
- 8$4-$ carbohydrate glucose, lactose , sucrose, starch are present. Different test is carried out with $4-$ carbohydratcs separately. Following observation takes place.View Solution
$(a)$ Heated : it is charred
$(b)$ $I_2$ solution : no violet colour
$(c)$ Fehling solution : reddish brown ppt. is observed
$(d)$ It does not reduce copper$(II)$ acetate to $Cu_2O$ . No red precipiated is observed in $5/ min$
The carboxylate which satisfy all the above observation
- 9View SolutionIncorrect statement from the following
- 10View SolutionWhich one of the following statements is correct?
