Download our appand get started for free
Similar Questions
- 1Vitamin ${B_{12}}$ contains metalView Solution
- 2The one that does not stabilize $2^{\circ}$ and $3^{\circ}$ structures of proteins isView Solution
- 3View SolutionThe central dogma of molecular genetics states that the genetic information flows from
- 4Which of the following base is found only in $RNA$ but not in $DNA$View Solution
- 5Consider the following reactions :View Solution
$(i)$ Glucose $+ ROH \quad \stackrel{\text { dry } HCl }{\longrightarrow}$ Acetal
$\xrightarrow[{{{\left( {C{H_3}CO} \right)}_2}O}]{{x\,eq.of}}$ acetyl derivative
$(ii)$ Glucose $\xrightarrow{{Ni/{H_2}}}A\xrightarrow[{{{\left( {C{H_3}CO} \right)}_2}O}]{{y\,\,eq.\,of}}$ acetyl derivative.
$(iii)$ Glucose $\xrightarrow[{{{\left( {C{H_3}CO} \right)}_2}O}]{{z\,ed.\,of}}$ acetyl derivative.
$' x ^{\prime},{ }^{\prime} y ^{\prime}$ and ${ }^{\prime} z^{\prime}$ in these reactions are respectively.
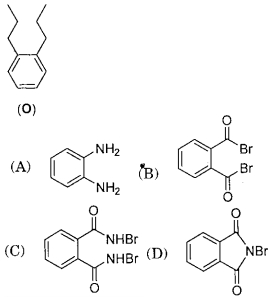
- 6Treatment of compound $O$ with $KMnO_4 / \mathrm{H}^{+}$gave $P$, which on heating with ammonia gave $Q$. The compound $Q$ on treatment with $Br_2 / NaOH$ produced $R$. On strong heating, $Q$ gave $S$, which on further treatment with ethyl $2$-bromopropanoate in the presence of $KOH$ followed by acidification, gave a compound $T$.View Solution
(image)
($1$) The compound $R$ is
(image)
($2$) The compound $T$ is
($A$) glycine
($B$) alanine
($C$) valine
($D$) serine
Given the answer question ($1$) and ($2$)
- 7View SolutionWhich of the following basic amino acid is least basic
- 8Which among the following compounds, is a $\beta$ ketohexo furanose?View Solution
- 9View SolutionGlucose in blood can be quantitatively determined with
- 10$4-$ carbohydrate glucose, lactose , sucrose, starch are present. Different test is carried out with $4-$ carbohydratcs separately. Following observation takes place.View Solution
$(a)$ Heated : it is charred
$(b)$ $I_2$ solution : no violet colour
$(c)$ Fehling solution : reddish brown ppt. is observed
$(d)$ It does not reduce copper$(II)$ acetate to $Cu_2O$ . No red precipiated is observed in $5/ min$
The carboxylate which satisfy all the above observation
