Which is not a reducing suger?
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionCanesugar on hydrolysis gives
- 2View SolutionWhich of the following statement is not correct?
- 3View SolutionWhich of the following vitamin is water soluble?
- 4View SolutionGlucose does not react with :-
- 5Assertion : Treatment of $D-$ glucose with dilute alkali affords an equilibrium mixture consisting of $D-$ mannose, $D-$ fructose and starting substance $D-$ glucose.View Solution
Reason : The reaction involves an intermediate in which hybridisation of $C_2$ changes from $sp^3$ to $sp^2$ - 6Molecular formula ${C_6}{H_{12}}{O_6}$ is ofView Solution
- 7View SolutionIn a linear tetrapeptide (Constituted with different amino acids), (number of amino acids) - (number of peptide bonds) is
- 8View SolutionAll of the following statements apply to proteins except
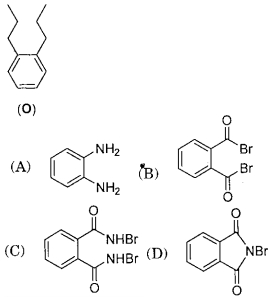
- 9Treatment of compound $O$ with $KMnO_4 / \mathrm{H}^{+}$gave $P$, which on heating with ammonia gave $Q$. The compound $Q$ on treatment with $Br_2 / NaOH$ produced $R$. On strong heating, $Q$ gave $S$, which on further treatment with ethyl $2$-bromopropanoate in the presence of $KOH$ followed by acidification, gave a compound $T$.View Solution
(image)
($1$) The compound $R$ is
(image)
($2$) The compound $T$ is
($A$) glycine
($B$) alanine
($C$) valine
($D$) serine
Given the answer question ($1$) and ($2$)
- 10View SolutionAmino acids are
