A massless spring $(k = 800\, N/m)$, attached with a mass $(500\, g)$ is completely immersed in $1\, kg$ of water. The spring is stretched by $2\, cm$ and released so that it starts vibrating. What would be the order of magnitude of the change in the temperature of water when the vibrations stop completely? (Assume that the water container and spring receive negligible heat and specific heat of mass $= 400\, J/kg\, K$, specific heat of water $= 4184\, J/kg\, K$)
JEE MAIN 2019, Diffcult
By law of conservation of energy
$\frac{1}{2} \mathrm{kx}^{2}=\left(\mathrm{m}_{1} \mathrm{s}_{1}+\mathrm{m}_{2} \mathrm{s}_{2}\right) \Delta \mathrm{T}$
$\Delta T=\frac{16 \times 10^{-2}}{4384}=3.65 \times 10^{-5}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe total energy of a particle, executing simple harmonic motion is
- 2Function $x$ = $A sin^2 wt + B cos^2 wt + C sin wt \ cos wt$ does not represents $SHM$ for this conditionView Solution
- 3The amplitude of a particle executing $SHM$ is $3\,cm$. The displacement at which its kinetic energy will be $25 \%$ more than the potential energy is: $.............cm$.View Solution
- 4The figure shows a graph between velocity and displacement (from mean position) of a particle performing $SHM\,:$View Solution
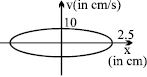
- 5A simple harmonic oscillator has an amplitude a and time period $T$. The time required by it to travel from $x = a$ to $x = \frac{a }{2}$ isView Solution
- 6A block of rectangular size of mass $m$ and area of cross-section $A$, floats in a liquid of density $\rho$. If we give a small vertical displacement from equilibrium, it undergoes $S H M$ with time period $T$, thenView Solution
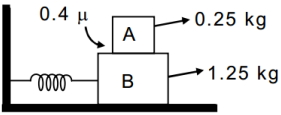
- 7Maximum amplitude(in $cm$) of $SHM$ so block A will not slip on block $B , K =100 N / m$View Solution
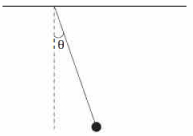
- 8A simple pendulum is released from rest at the horizontally stretched position. When the string makes an angle $\theta$ with the vertical, the angle $\phi$ which the acceleration vector of the bob makes with the string is given byView Solution
- 9A particle executes $S.H.M.$ with a period of $6$ second and amplitude of $3\, cm$. Its maximum speed in $cm/sec$ isView Solution
- 10View SolutionWhich of the following function represents a simple harmonic oscillation
