Question Bank
Explore our large set of questions to practice for your standard seamlessly- 1નીચેની પ્રક્રિયામાં $Y$ શું હશે ?View Solution
${C_2}{H_2}\xrightarrow{{{O_3}}}X\xrightarrow{{Zn/C{H_3}COOH}}Y$
- 2નીચેની બંધ વિયોજન ઉર્જાઓનો ક્રમ કયો છે ?View Solution
$I. \,CH_3 - H$
$II.\, CH_3CH_2-H$
$III.\, CH_2 = CH - CH_2-H$
$IV.\, C_6H_5$
- 3નીપજ $(C)$ શું હશે ?View Solution
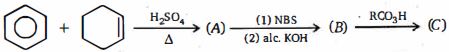
- 4પદાર્થનું અણુસૂત્ર છે તો $X$ પદાર્થની સંખ્યા કેટલી થાય ?View Solution
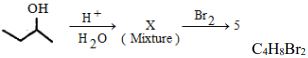
- 5View Solutionપોટેશિયમ એસિટેટના વિધુતવિભાજનથી ......... મળે છે.
- 6પ્રકાશીય સક્રિય હાઈડ્રોકાર્બન $(X)$ ઉપર ઉદ્દીપકીય હાઈડ્રોઝીનેશન કરતા પ્રકાશીય નિષ્ક્રિય પદાર્થ $(Y), C_6H_{14}$ મળે છે. હાઈડ્રોકાર્બન $(X)$ એ કયો છે ?View Solution
- 7પ્રકિયા $CH_2 = CH_2 + Br_2$ $\xrightarrow{{Na{I_{\left( {aq} \right)}}}}$ માટે નીચેનામાંથી ક્યુ વિધાન સાચું નથી ?View Solution
- 8View Solutionપ્રકિયા ની નીપજ કઈ હશે ?
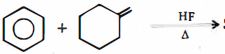
- 9પ્રકીયક $(A)$ શું હશે ?View Solution
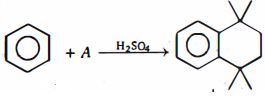
- 10પ્રક્રિયા $(I)$ અને $(II)$ માં મુક્ત થતા વાયુઓ.......છે.View Solution
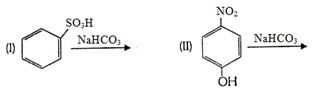
- 11પ્રક્રિયામાં $'X' $ શું છે ?View Solution
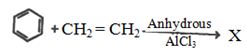
- 12પ્રક્રિયામાંં $C{H_3}C{H_2}CH\, = \,\,C{H_2}\,\,\mathop {\xrightarrow{{1.\,Hg{{(OAc)}_2}.\,{H_2}O}}}\limits_{2.\,NaB{D_4}} $ નીપજ કઈ છે ?View Solution
- 13View Solutionપ્રક્રિયામાં અંતિમ નિપજ = .......
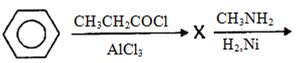
- 14View Solutionપ્રક્રિયામાં ઉદભવતી નીપજ કઈ છે ?
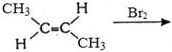
- 15View Solutionપ્રક્રિયામાં કયો આલ્કીન મુખ્ય નીપજ તરીકે મળે છે ?
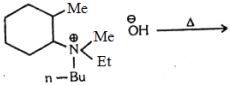
- 16પ્રક્રીયામાંView Solution
$C{{H}_{3}}CH\,=\,\,C{{H}_{2}}\,+\,\,{{H}_{2}}O\,\,+\,\,[O]\,\,\underset{Acid}{\mathop{\xrightarrow{KMn{{O}_{4}}}}}\,$$\begin{array}{*{20}{c}}
{C{H_3} - CH - C{H_2}} \\
{\,\,\,\,\,\,\,\,\,|\,\,\,\,\,\,\,\,\,\,\,\,\,|} \\
{\,\,\,\,\,\,\,\,OH\,\,\,\,\,\,\,OH}
\end{array}$ $\xrightarrow{{[O]}}{\mkern 1mu} {\mkern 1mu} X{\mkern 1mu} {\mkern 1mu} + {\mkern 1mu} {\mkern 1mu} HCOOH$$X$ શું છે ?
- 17પ્રોપાઇનની પ્રવાહી $H_2SO_4$ સાથેની પ્રક્રિયા $HgSO_4$ ની હાજરીમાં કરતા મુખ્ય નીપજ કઇ મળે છે?View Solution
- 18પ્રોપાઇનનુ $SeO_2$ દ્વારા ઓક્સિડેશન કરતા .... મળે છે.View Solution
- 19પ્રોપિનની $ICl$ સાથેની પ્રક્યિાથી મળતી નીપજ ............. છે.View Solution
- 20ફ્રિડલ ક્રાફટસ આલ્કીલેશનમાં $AlCl_3$ સાથે વપરાતો બીજો યોગ્ય પદાર્થ.......... છે.View Solution
- 21View Solutionફ્રિડલ-ક્રાફ્ટ પ્રક્રિયામાં નિર્જળ એલ્યુમિનયમ ક્લોરાઇડ વપરાય છે, કારણ કે તે ............
- 22View Solutionબતાવેલ તમામ હાઇડ્રોકાર્બન ખૂબ નબળા એસિડ્સ છે. એક જોકે અન્ય કરતા વધુ એસિડિક છે. કયું એક સૌથી મજબૂત એસિડ છે
- 23View Solutionબંધ ટયૂબમાં મિથાઇલ ક્લોરાઇડને ઝિંક સાથે ગરમ કરતા............... ઉત્પન્ન થાય છે.
- 24View Solutionબેન્ઝિન ........ દ્વારા મળે છે.
- 25બેન્ઝિનની $AlCl_3$ ની હાજરીમાં $CH_3COCl$ સાથેની પ્રક્રિયાથી ........... મળે છે.View Solution
- 26બેન્ઝિનની જલદ $HNO_3$ અને જલદ $H_2SO_4$ સાથે $353$ થી $363 \,K$ તાપમાને પ્રક્રિયા કરતાં મળતી નીપજ.... છે.View Solution
- 27બેન્ઝિનની સાંદ્ર $HNO_3$ અને સાંદ્ર $H_2SO_4$ ના મિશ્રણ સાથે પ્રક્રિયા કરી ત્યારબાદ $Cl_2 / FeCl_3$ સાથેની પ્રક્રિયા ... આપે છે.View Solution
- 28View Solutionબેન્ઝિનનુ ટોલ્યુઇનમાં રૂપાંતર ......... દ્વારા થાય છે.
- 29બેન્ઝિનમાં બધા જ કાર્બનો વચ્ચે $C - C$ બંધ લંબાઈ સમાન છે કારણ કે...View Solution
- 30View Solutionબેન્ઝિન યોગશીલ પ્રક્રિયા સરળતાથી આપતું નથી કારણ કે ...............
- 31બ્રોમિનેશનની પ્રક્રિયામાં $1\,^oH, 2\,^oH$ અને $3\,^oH$ માં પ્રક્રિયાશીલતાનો પ્રક્રિયા દર અનુક્રમે $1 : 82 : 1600$ છે તો પ્રક્રિયામાં $ (a)$ અને $(b)$ નીપજ કેટલી ટકાવારીમાં ઉપજ મળશે ?View Solution
$C{{H}_{3}}\,-\,\,\overset{\overset{C{{H}_{3}}}{\mathop{|}}\,}{\mathop{C}}\,H\,\,-\,\,C{{H}_{3}}(excess)\,\,+\,\,B{{r}_{2}}\,\xrightarrow{hv}$
$\,\,\,\,C{{H}_{3}}\,-\,\,\overset{\overset{C{{H}_{3}}}{\mathop{|}}\,}{\mathop{\underset{\underset{Br}{\mathop{|}}\,}{\mathop{C}}\,}}\,\,\,-\,\,C{{H}_{3}}\,+\,\,C{{H}_{3}}\,-\,\,CH\,\,-\,\,C{{H}_{2}}\,-\,\,Br$
- 32બ્રોમોઆલ્કેનને સોડીયમ એમાઇડ અને આલ્કીનના મિશ્રણમાં ભેળવીને $3-$ઓકટાઇન મેળવવામાં આવે છે. તો આ બ્રોમો આલ્કીન અને આલ્કાઇન અનુક્રમે કયા હશે ?View Solution
- 33View Solutionમિથાઇલ મેગ્નેશિયમ બ્રોમાઇડની ઇથાઇલ આલ્કોહોલ સાથેની પ્રક્રિયાથી મળતી નીપજ .......છે.
- 34View Solutionમિથેન અને ક્લોરિનની પ્રકિયા મિશ્રણમાં ઓક્સિજનનો ઉમેરો ............... (photochemical chlorination)
- 35મિથેનોલના દ્રાવણમાં ઇથિલિનની બ્રોમિન સાથેની પ્રક્રિયાથી $1, 2-$ ડાયબ્રોમો ઇથેન ઉપરાંત $Br-CH_2CH_2OCH_3$ પણ મળે છે. કારણ કે ......View Solution
- 36લિથિયમ ડાય $-૩-$ પેન્ટાઇલ ક્યુપ્રેટની ઇથાઇલ બ્રોમાઇડ સાથેની પ્રક્રિયાથી $C_7H_{16}$ આણ્વિય સૂત્ર ધરાવતો આલ્કેન મળે તો આલ્કેનનું બંધારણ ............ થશે.View Solution
- 37વધારે હાઈડ્રોજન વાયુ સાથે નેફ્થાલિન $(P)$ નું હાઇડ્રોજન $1, 2, 3,4-$ ટેટ્રાહાઇડ્રોનેફ્થાલિન $(Q).$ પર શુધ્ધ રીતે અટકે છે. આ પ્રયોગથી શું નિષ્કર્ષ કાઢીશકાય છે?View Solution
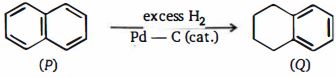
- 38View Solutionશુદ્ધ મિથેન ................ દ્વારા ઉત્પન્ન કરી શકાય છે.
- 39સંયોજન $Y$ આપવા માટે પાણી સાથે ચૂનો સાથે પ્રક્રિયા આપતા સંયોજન $X$એ એક ગરમ કોક બનાવ્યો જે $873$ પર લાલ ગરમ આયર્ન પર પસાર થતાં $Z$ ઉત્પન્ન કરે છે તો સંયોજન $Z$ શું હશે ?View Solution
- 40View Solutionસાયક્લોપેન્ટાડાઈન સાયક્લોપેંટેન કરતા વધુ એસિડિક છે. કારણ કયું છે
- 41સાયક્લોહેકઝેનની હાઇડ્રોજીનેશન ઉષ્મા $28.6 \,kcal$ છે અને સાયકલોહેકઝાડીઈન કરતા થોડું બમણું છે, $(55.4\,kcal)$ તો બેન્ઝીનના હાઇડ્રોજીનેશન ઉષ્મા કેટલી થશે ? જે ત્રણ દ્વિબંધ ધરાવે છે ?View Solution
- 42View Solutionસિલ્વર પાઉડરની ક્લોરોફોર્મ સાથેની પ્રક્રિયાથી .... મળે છે.
- 43સિસ $-2-$ બ્યુટિનમાં $Br_2$ ઉમેરતા ........... મળે છે.View Solution
- 44સીસ$-2-$બ્યુટીનની $CCl_4$ માં $Br_2$ ની સાથે પ્રક્રિયા કરતા મુખ્યત્વે શું બનાવે છે ?View Solution
- 45View Solutionસૂર્યપ્રકાશની હાજરીમાં મિથેન અને ક્લોરિન વચ્ચેની પ્રક્રિયાથી કઇ નીપજ મળતી નથી ?
- 46નીચેનામાંથી ક્યાં સંયોજનને પ્રબળ એસિડ સાથે ગરમ કરતાં $2,{3-}$ડાયમિથાઇલ$-{2-}$બ્યુટીન બનાવી શકાય છે?View Solution
- 47નીચેના પૈકી ક્યો પ્રક્રિયક $1-$બ્યુટાઇન અને $2-$બ્યુટાઇન વચ્ચેનો ભેદ પારખી શકશે ?View Solution
- 48નીચેની પ્રક્રિયાની મુખ્ય નીપજ કઈ છે ?View Solution
$\begin{array}{*{20}{c}}
{C{H_3}\,\,\,\,\,\,\,} \\
{|\,\,\,\,\,\,\,\,\,\,\,\,\,} \\
{{H_3}C - C - CH = C{H_2}} \\
{|\,\,\,\,\,\,\,\,\,\,\,\,\,} \\
{C{H_3}\,\,\,\,\,\,\,\,\,}
\end{array}$ $\xrightarrow{{{H_2}O/{H^ \oplus }}}{\mkern 1mu} \mathop A\limits_{Major\,product} \, + \,\mathop B\limits_{Minor\,product} $ - 49$FeCl_3$ની હાજરીમાં $Cl_2$ સાથે ટોલ્યુઇનની પ્રક્રિયા $X$ આપે છે અને પ્રકાશની હાજરીમાં પ્રક્રિયા $Y$ આપે છે આમ, $X$ અને $Y$ છે,...View Solution
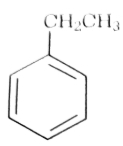
- 50પ્રક્રિયાઓના સમૂહમાં, ઇથાઇલબેન્ઝિનનું ઉત્પાદન $D$ સ્વરૂપે થાય છેView Solution
(image) $\xrightarrow[{KOH}]{{KMn{O_4}}}\,B\,\xrightarrow[{FeC{L_3}}]{{B{r_2}}}\,C\,\xrightarrow[{{H^ + }}]{{{C_2}{H_5}OH}}\,D$
$D$ શું હશે?
- 51View Solutionપ્રવાહી હાઇડ્રોકાર્બનનું વાયુરૂપ હાઇડ્રોકાર્બનમાં રૂપાંતર કઈ રીતે થઈ શકે છે ?
- 52$CH_3Cl$ સાથે બેન્ઝિન નિર્જળ $AlCl_3$ની હાજરીમાં પ્રક્રિયા કરીને શું બનાવે છે?View Solution
- 53View Solutionનીચેનામાંથી કયું સંયોજન સિસ- ટ્રાન્સ (ભૌમિતિક) સમઘટકતાનું પ્રદર્શન કરશે?
- 54હાઇડ્રોકાર્બનમાં $C_2, C_3, C_5$ અને $C_6$ સંકરણની અવસ્થા જણાવો,View Solution
$\begin{array}{*{20}{c}}
{C{H_3}\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,C{H_3}\,\,\,\,\,\,\,} \\
{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,|\,\,\,\,\,\,\,\,\,\,\,\,} \\
{\mathop C\limits_7 {H_3} - \mathop C\limits_6 - \mathop C\limits_5 H = \mathop C\limits_4 H - \mathop C\limits_3 H - \mathop C\limits_2 \equiv \mathop C\limits_1 H} \\
{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,} \\
{C{H_3}\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}
\end{array}$જે નીચેના ક્રમમાં છે.
- 55$\begin{array}{*{20}{c}}View Solution
{{H_3}C - CH - CH = C{H_2}} \\
{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,} \\
{C{H_3}\,\,\,\,\,\,\,\,\,\,\,\,\,}
\end{array}$ $+HBr \rightarrow A$પ્રક્રિયામાં $A$(મુખ્યત્વે) ...... હશે.
- 56હાઇડ્રોકાર્બનમાં ,View Solution
$\mathop C\limits_6 {H_3} - \mathop C\limits_5 H = \mathop C\limits_4 H - \mathop C\limits_3 {H_2} - \mathop C\limits_2 \equiv \mathop C\limits_1 H$
કાર્બન્સ $1, 3$ અને $5$ ના સંકરણની સ્થિતિ નીચેના ક્રમમાં છે, તો સાચો ક્રમ શોધો.
- 57બ્યુટાઇલ$-1$ ની નીચેની પ્રક્રિયામાં મળતી નીપજ $C$ જણાવો.View Solution
$CH_3CH_2-C \equiv CH+ HCl \rightarrow B \xrightarrow{{HI}} C$
- 58$C_5H_{10}$ અણુસૂત્ર ધરાવતુ નીચેના પૈકી ક્યુ સંયોજન ઓઝોનોલિસિસથી એસિટોન આપશે?View Solution
- 59નીચેની પ્રક્રિયાની નિપજો કઈ છે ?View Solution
$C{H_3}C \equiv \,C\,C{H_2}C{H_3}\mathop {\xrightarrow[{(2)\;Hydrolysis}]{}}\limits^{(1)\;\;{O_3}} $ .......
- 60નિર્જળ $AlC{l_3}$ નો ઉદ્દીપક તરીકે ઉપયોગ કરીને, નીચેના પૈકી કઇ પ્રક્રિયા ઇથાઇલ બેન્ઝિન $(PhEt)$ આપશે ?View Solution
- 61પેરોક્સાઇડની હાજરીમાં પ્રોપિનની $HBr$ સાથેની પ્રક્રિયા શુ આપશે ?View Solution
- 62View Solutionનીચે પૈકી કોની મુક્ત મુલક વિસ્થાપન પ્રક્રિયા છે?
- 63સંયોજન $\begin{array}{*{20}{c}}View Solution
{C{H_{3\,\,}}\,\,\,\,\,\,\,} \\
{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,} \\
{C{H_3} - C = CH - C{H_3}}
\end{array}$ એ $KMn{O_4}$ ની હાજરીમાં $NaI{O_4}$ સાથે પ્રક્રિયામાં દ્વારા શું આપશે ? - 64View Solutionનીચેનામાંથી કોનો ઉપયોગ ઇથેનથી ઇથેનને અલગ કરવા માટે થતો નથી?
- 65View Solutionનીચેની કઈ પદ્ધતિ વડે, પ્રોપીનમાંથી પ્રોપેન મેળવવામાં આવે છે
- 66પ્રક્રિયા $C{H_2} = C{H_2} + {H_2}\mathop {\xrightarrow{{Ni}}}\limits_{250 - {{300}\,^o}C} C{H_3} - C{H_3}$ને કહેવાય છે..... .View Solution
- 67View Solutionપ્રોપીનમાંથી પ્રોપેન કઈ પદ્ધતિ દ્વારા મેળવવામાં આવે છે?
- 68નીચેનામાંથી કયો $C - H$ બંધ સૌથી નીચી બંધ વિયોજન ઊર્જા ધરાવે છે?View Solution
- 69View Solutionજંતુનાશક ગૈમેક્સેનનું રાસાયણિક નામ શું છે?
- 70ફ્રિડલ-ક્રાફ્ટ પ્રક્રિયા દ્વારા ટોલ્યુઇનની બનાવટમાં, $AlC{l_3}$ ની હાજરીમાં વપરાતા પ્રક્રિયકો જણાવો.View Solution
- 71પ્રક્રિયા એ $C{H_4} + C{l_2} \xrightarrow{uv \,light} \mathop {}\limits_{} C{H_3}Cl + HCl$ નું એક ઉદાહરણ છેView Solution
- 72View Solutionઆમાંથી કયા એરિન્સ સાથે સુસંગત નથી
- 73નીચેની પ્રક્રિયાની શ્રેણીમાં નીપજ $D$ ઓળખોView Solution
$C{H_3}COOH\xrightarrow{{LiAl{H_4}}}A\mathop {\xrightarrow{{{H^ + }}}}\limits_{443\,K} B\xrightarrow{{B{r_2}}}C\mathop {\xrightarrow{{alc.}}}\limits_{KOH} D$
- 74View Solutionઇથાઇનમાં ત્રિ-બંધ કોનાથી બને છે અથવા આલ્કાઈનનું નળાકાર આકાર કોના કારણે છે?
- 75View Solutionવ્યાપારિક ગેસોલિનમાં હાઇડ્રોકાર્બનનો ક્યો પ્રકાર વધુ ઇચ્છનીય છે ?
- 76View Solutionનીચેના પૈકી કઇ પ્રક્રિયામાં સારા પ્રમાણમાં હાઇડ્રોકાર્બન નીપજ મળવાની અપેક્ષા રાખી શકાય ?
- 77નીચેની પ્રક્રિયામાં $M$ અને $R$ અનુક્રમે ........ હશે.View Solution
$C{H_2} = C{H_2}\xrightarrow[{oxid}]{{Hypochloro}}$ $M\xrightarrow{R}\begin{array}{*{20}{c}}
{C{H_2} - OH} \\
{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,} \\
{C{H_2} - OH}
\end{array}$ - 78$FeC{l_3}$ ની હાજરીમાં બેન્ઝિનના ક્લોરીનેશનના કિસ્સામાં ઇલેક્ટ્રોન અનુરાગી કોણ છે?View Solution
- 79$s{p^3}$ વર્ણસંકરિત કાર્બન અણુ અને અન્ય કાર્બન અણુ વચ્ચે બંધ લંબાઈ ઓછામાં ઓછી ........ છે.View Solution
- 80નીચેની પ્રક્રિયા કઈ પ્રક્રિયા છે?View Solution
$C{H_2} = CH - C{H_3} + HBr \to C{H_3}CHBr - C{H_3}$
- 81View Solutionકયુ અપસ્ફોટનવિરોધી સંયોજન શ્રેષ્ઠ છે અથવા નીચેનામાંથી કોઈ એક પદાર્થ અપસ્ફોટનવિરોધી સંયોજન તરીકે ઉપયોગમાં લેવાય છે?
- 82$3, 3-$ ડાયમિથાઇલ $-2-$બ્યુટેનોલને ${H_2}S{O_4}$ સાથે ગરમ કરતા મળતી મુખ્ય નીપજ કઇ હશે ?View Solution
- 83આલ્કીન $R - C - H = C{H_2}$ ની ${B_2}{H_{6}}$ સાથેની પ્રક્રિયાથી નીપજ $B$ મળે છે, જેનું આલ્કલાઇન હાઇડ્રોજન પેરોક્સાઇડ વડે ઓક્સિડેશન શુ ઉત્પન્ન કરશે ?View Solution
- 84બેન્ઝિનનું નાઇટ્રેશન એ હુમલો અથવા પ્રતિક્રિયાશીલ અથવા ઇલેક્ટ્રોન અનુરાગી છે અથવા બેન્ઝિનના નાઇટ્રેશનમાં સાંદ્ર $HN{O_3}$ અને ${H_2}S{O_4}$ સાથે રિંગ પર હુમલો કોના દ્વારા કરવામાં આવે છેView Solution
- 85View Solutionબેન્ઝિન માટે નીચેનામાંથી સાચુ વિધાન પસંદ કરો.
- 86View Solutionનીચેના સંયોજનો પૈકી એક, જે કેન્દ્રાનુરાગી નાઇટ્રેશન તરફ સૌથી વધુ ક્રિયાશીલ છે તે છે...
- 87View Solutionકેલ્શિયમ કાર્બાઇડ સાથે પાણી પ્રક્રિયા કરતા શું આપશે?
- 88ફ્રીડલ-ક્રાફ્ટની પ્રક્રિયામાં નિર્જળ $AlC{l_3}$ નો ઉપયોગ થાય છે કારણ કે તે છે.... .View Solution
- 89બેન્ઝિન $AlC{l_3}$ની હાજરીમાં $C{H_3}COCl$ સાથે પ્રક્રિયા કરીને શું આપશે?View Solution
- 90View Solutionઇથિલીનમાં ક્યાં પ્રકારનું સંકરણ હોય છે ?
- 91$2, 3-$ ડાઇબ્રોમો $-3-$ મિથાઇલ પેન્ટેન ઝિંક પાવડરથી ગરમ થયા બાદ પરિણામી નીપજ અલગ કરવામાં આવે છે અને ફૉસ્ફરસની હાજરીમાં $HI$ સાથે ગરમ કરતાં પ્રક્રિયામાં બનતી અંતિમ નીપજનું બંધારણ શું હશે?View Solution
- 92$KMn{O_4}$ની લાક્ષણિકતા જાંબુડિયા રંગની આલ્કિન સાથે તેની પ્રક્રિયામાં અદશ્યતા થવું તે અસંતૃપ્ત કસોટી છે,તે ..... તરીકે ઓળખાય છેView Solution
- 93$R - C{H_2} - CC{l_2} - R \xrightarrow{{{\text{Reagent}}}}R - C \equiv C - R$View Solution
$reagent$ શું હશે?
- 94View Solutionએસિટિલિનીક હાઇડ્રોજન એસિડિક હોય છે, કારણ કે ........
- 95ચક્રીય હાઇડ્રોકાર્બન જેમાં બધા કાર્બન અને હાઇડ્રોજન પરમાણુ એક જ સમતલમાં હોય છે. બધા કાર્બન-કાર્બન બંધો સમાન લંબાઈના અને $1.54\,\mathop A\limits^o $ કરતાં ઓછા છે, પરંતુ $1.34\,\mathop A\limits^o $ કરતાં વધુ લંબાઈના હોય છે ,તો બંધ કોણ હશે?View Solution
- 96View Solutionજ્યારે એમોનીયામય કયુપ્રસ ક્લોરાઇડ સાથે પ્રક્રિયા કરવામાં આવે છે, જે નીચેનામાંથી કોણ કોપર વ્યુત્પન્ન રચે છે?
- 97જ્યારે પ્રોપિન પેરોક્સાઇડ્સની હાજરીમાં $HBr$ સાથે પ્રક્રિયા આપે છે ત્યારે મુખ્ય નીપજ કઈ રચાય છે?View Solution
- 98નિર્જળ ઝિંક ક્લોરાઇડની હાજરીમાં $2-$મિથાઇલ પ્રોપીન સાથે એસિટાઇલ ક્લોરાઇડથી ગરમ કરતાં કાર્બનિક સંયોજન નિર્માણ પામે છે,તેનું સૂત્ર શું હશે?View Solution
- 99નીચેનામાંથી જે સૌથી મજબૂત $o, p-$ નિર્દેશક સમૂહ છે .....View Solution
- 100સંયોજન $(iii)$ ને બાકીના સંયોજનોથી અલગ ઓળખવા માટે નીચેના પૈકી સૌથી યોગ્ય પ્રક્રિયક ક્યો છે ?View Solution
$(i)$ $C{H_3} - C \equiv C - C{H_3}$
$(ii)$ $C{H_3} - C{H_2} - C{H_2} - C{H_3}$
$(iii)$ $C{H_3} - C{H_2} - C \equiv CH$
$(iv)$ $C{H_3} - CH = C{H_2}$