Proteins do not respond to
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionType of amino acids obtained by hydrolysis of proteins is :
- 2A sequence of how many nucleotides in messenger $RNA$ makes a codon for an amino acidView Solution
- 3The structure of a peptide is given belowView Solution
If the absolute values of the net charge of the peptide at $pH =2, pH =6$, and $pH =11$ are $\left| z _1\right|,\left| z _2\right|$ and $\left|z_3\right|$, respectively, then what is $\left|z_1\right|+\left|z_2\right|+\left|z_3\right|$ ?
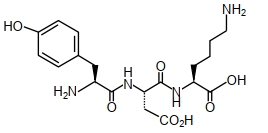
- 4View SolutionWhich of the following is water soluble vitamin ?
- 5View SolutionAnimal starch is the other name of.
- 6View SolutionWhich one of the following is a peptide hormone ?
- 7The binding of oxygen by haemoglobin $(Hb)$ forming $(HbO_2)$, is partially regulated by the concentration of $H_3O^+$ and dissolved $CO_2$ in blood.View Solution
$Hb{O_2} + {H_3}{O^ + }C{O_2} \rightleftharpoons \left( {{H^ + } - Hb - C{O_2}} \right) + {O_2} + {H_2}O$
Release of $O_2$ is favoured when there is
- 8$DNA$ multiplication is calledView Solution
- 9View SolutionThe nucleic acid base having two possible binding sites is
- 10View SolutionWhich of the following is not possible in glucose?
