Download our appand get started for free
Similar Questions
- 1Number of compounds from the following which will not produce orange red precipitate with Benedict solution is $..............$. Glucose, maltose, sucrose, ribose,$2-$deoxyribose, amylose, lactose.View Solution
- 2Which of the following statement($s$) is(are) true ?View Solution
$(1)$ Oxidation of glucose with bromine water gives glutamic acid
$(2)$ The two six-membered cyclic hemiacetal forms of $D-(+)$-glucose ard called anomers
$(3)$ Hydrolysis of sucrose gives dextrorotatory glucose and laevorotatory fructose
$(4)$ Monosaccharides cannot be hydrolysed to give polyhydroxy aldehydes and ketones
- 3View SolutionAn example of a disaccharide made up of two units of the same monosaccharides is
- 4The $pK_a$ values for the three ionizable groups $X , Y$ and $Z$ of glutamic acid are $4.3, 9.7$ and $2.2$ respectivelyView Solution
$\begin{matrix}
\underset{X}{\mathop{H}}{{O}_{2}}C-C{{H}_{2}}-CH-C{{O}_{2}}\underset{Z}{\mathop{H}}\, \\
\,\,\,\,\,\,\,\,\,\,\,| \\
\,\,\,\,\,\,\,\,\,\,\,\,\,+N{{H}_{3}} \\
\end{matrix}$The isoelectric point for the amino acid is
- 5Choose the peptide that matches the abbreviation $Phe-Val-Ala.$View Solution
- 6View SolutionOn complete hydrolysis of starch, we finally get
- 7View SolutionBlood calcium level can be increased by the administration of
- 8View SolutionA nanopeptide contains …….. peptide linkages
- 9View SolutionEnzymes belong to which class of compounds
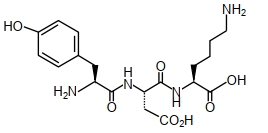
- 10The structure of a peptide is given belowView Solution
If the absolute values of the net charge of the peptide at $pH =2, pH =6$, and $pH =11$ are $\left| z _1\right|,\left| z _2\right|$ and $\left|z_3\right|$, respectively, then what is $\left|z_1\right|+\left|z_2\right|+\left|z_3\right|$ ?